Yu Katayama
Position: Associate Professor
Degree: Ph.D. (Engineering)
Laboratory: Energy Materials Science
to Laboratory Homepage
Design and Analysis of the Electrode-Electrolyte Interface
(Electro)catalysis provides exciting opportunities to address some of the impending global changes of the 21st century ranging from energy and environment to food and chemical production. The unique catalytic activity of the electrode stems from its distinctive geometric and electronic structure, which can be tuned to facilitate numerous (electro)catalytic processes on the surface, including insertion, adsorption, conversion, and desorption of reactant and product molecules in organic/aqueous media. Although the electrocatalytic activity has been improved by tuning the electronic structure of the electrode materials, there has been little focus on engineering the electrode/electrolyte interface. Furthermore, little is known about molecular processes and interactions that govern reaction efficiency, activity, stability, and selectivity, limiting the design of new functional electrode/electrolyte interfaces. Therefore, there are immense opportunities for fundamental research of electrode/electrolyte interfaces to identify governing parameters that dictate the complex interactions at interfaces. Our group aims to connect the understanding of surface reaction mechanisms obtained by in situ/ex situ spectroscopic techniques combined with theoretical analysis to activity and stability of the catalysts, as well as to establish novel design principles for designing electrode/electrolyte interfaces. 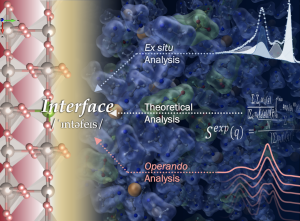
Revolutionize the Electrochemical “Power-to-X” Technology
With the emergence of abundant renewable electricity, industries are replacing conventional thermochemical routes with electrification to reduce carbon intensity. However, electrochemical material conversion, so-called Power-to-X technology, faces challenges, as traditional thermochemical ways are often not easily electrified. Furthermore, highly active and state-of-the-art, high cost and scarcity of noble metal-based catalysts necessitate the development of alternative (electro)catalysts. Therefore, we are developing precious metal-free catalysts for efficient electrocatalytic reactions to synthesize chemical fuels and feedstocks using renewable electricity, abundant resources, and water. Our group seeks to apply this approach to a variety of different chemistries where electrochemical routes are promising, such as the conversion of carbon dioxide into chemical feedstocks (e.g., methane), water electrolysis to convert water into hydrogen and oxygen, and electrochemical ammonia synthesis that can replace energy-consuming conventional Haber-Bosch Process. Our studies on electrocatalysts encompass extensive experimental components, e.g., the operando investigation of surface and interfacial processes using advanced characterization techniques. In addition, our group aims to integrate the fundamental understanding of catalyst materials into industrially relevant power-to-X systems. 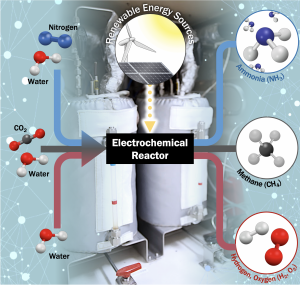
Exploring the Novel Reactions in the “Electro-Nanoreactor”
Beyond bulk metal catalysts, we have employed host-guest layered materials as platforms to design electrocatalysts that combine the tunability of molecular catalysts with the practical advantages of heterogeneous catalysts. Host-guest layered materials are a class of hybrid materials constructed from metal oxide layer (host layer) and compound with cationic nature (guest layer). Compared with traditional heterogeneous electrocatalysts such as metal alloys, host-guest layered materials possess unprecedented tunability by modifying components within the guest layer. Furthermore, we have demonstrated that the guest layer can contain metal complexes, sub-nano/single atom metals, and hydrated cations, which can be utilized as the electrocatalyst by the electron transfer through host metal oxides. Therefore, the guest layer can be considered a tunable electrochemical nanoreactor, containing reactant and electrocatalysts. Future works will focus on the design of the electro-nanoreactor, which is then implemented in a variety of electrocatalysis, including oxygen reduction reaction, water splitting, and CO2 reduction reaction. These experiments fit together to make a breakthrough in electrocatalysis to address critical energy problems across the globe. 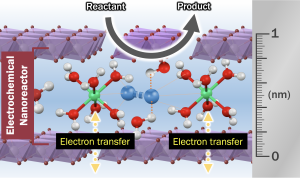
Message to Students
We work closely with researchers worldwide to develop novel materials/devices to solve global environmental/energy challenges. We welcome motivated students across the globe with strong academic backgrounds in electrochemistry, materials science, chemical engineering, or closely related fields.
Publication
- Botao Huang, Reshma R. Rao, Sifan You, Kyaw Hpone Myint, Yizhi Song, Yanming Wang, Wendu Ding, Livia Giordano, Yirui Zhang, Tao Wang, Sokseiha Muy, Yu Katayama, Jeffrey C. Grossman, Adam P. Willard, Kang Xu, Ying Jiang, Yang Shao-Horn, Cation- and pH-Dependent Hydrogen Evolution and Oxidation Reaction Kinetics, Journal of the American Chemical Society Au, 1, 1674−1687 (2021). https://doi.org/10.1021/jacsau.1c00281
- Reshma R Rao, Manuel J Kolb, Livia Giordano, Anders Filsøe Pedersen, Yu Katayama, Jonathan Hwang, Apurva Mehta, Hoydoo You, Jaclyn R Lunger, Hua Zhou, Niels Bendtsen Halck, Tejs Vegge, Ib Chorkendorff, Ifan E.L. Stephens, Yang Shao-Horn, Operando Identification of Site-Dependent Water Oxidation Activity on Ruthenium Dioxide Single-Crystal Surfaces, Nature Catalysis 3, 516–525 (2020). https://doi.org/10.1038/s41929-020-0457-6
- Yirui Zhang, Yu Katayama (co-first author), Ryoichi Tatara, Livia Giordano, Yang Yu, Dimitrios Fraggedakis, Jame G. Sun, Filippo Maglia, Roland Jung, Martin Z. Bazant, Yang Shao-Horn, Revealing Electrolyte Oxidation via Carbonate Dehydrogenation on Ni-based Oxides in Li-ion Batteries by In Situ Fourier Transform Infrared Spectroscopy, Energy & Environmental Science 13, 183–199 (2020). https://doi.org/10.1039/C9EE02543J
- Riho Matsuoka, Masayuki Shibata, Kousuke Matsuo, Ryansu Sai, Hiromori Tsutsumi, Kenta Fujii, Yu Katayama, Importance of Lithium Coordination Structure to Lithium-Ion Transport in Polyether Electrolytes with Cyanoethoxy Side Chains: An Experimental and Theoretical Approach, Macromolecules, 53, 9480−9490 (2020). https://doi.org/10.1021/acs.macromol.0c01634
- Jonathan Hwang, Reshma R. Rao, Livia Giordano, Yu Katayama, Yang Yu, Yang Shao-Horn, Perovskites in Catalysis and Electrocatalysis, Science 358, 751–756 (2017). https://doi.org/10.1126/science.aam7092
- Yu Katayama, Takeou Okanishi, Hiroki Muroyama, Toshiaki Matsui, Koichi Eguchi, Enhanced Supply of Hydroxyl Species in CeO2-Modified Platinum Catalyst Studied by in Situ ATR-FTIR Spectroscopy, ACS Catalysis 6, 2026–2034 (2016). https://doi.org/10.1021/acscatal.6b00108